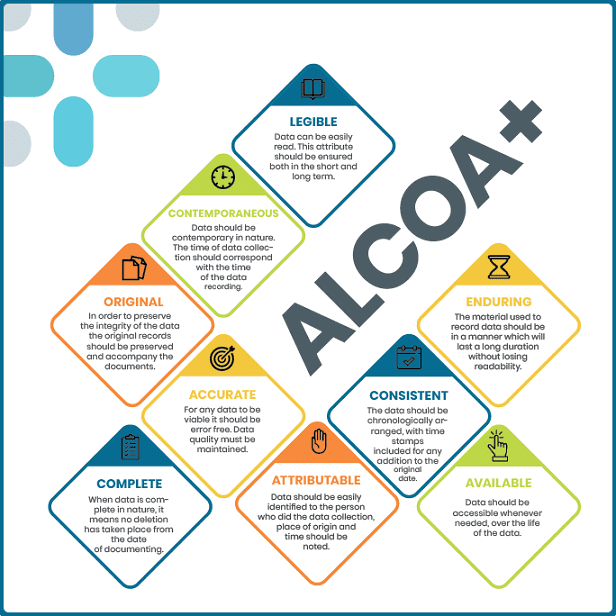
ALCOA+ is a quality management framework used in various industries, particularly in healthcare and clinical research. It is an extension of the original ALCOA principles, which are a set of guidelines to ensure data integrity in regulated environments. ALCOA stands for:
- Attributable: Data should be linked to the person responsible for its creation or modification.
- Legible: Information should be recorded in a clear and readable format.
- Contemporaneous: Data should be recorded at the time it is generated or observed.
- Original: The original record should be preserved, with any subsequent changes documented.
- Accurate: Data should be free from errors, complete, and correct.
The “+” in ALCOA+ represents additional principles that enhance data integrity. These principles include:
- Primary: Data should be captured directly from the source or be a verified copy.
- Data Integrity: Records should be protected against unauthorized changes and tampering.
- + refers to any additional requirements specified by relevant regulatory or industry standards.
Adhering to the ALCOA+ framework helps organizations ensure the reliability and trustworthiness of their data by maintaining accurate, complete, and unalterable records. By following these principles, organizations can improve data quality, transparency, and compliance with regulatory requirements.